Microfluidics Symposium: Innovations in Sensors, Materials, and Smart Microsystems
Abstracts
Session I
Presentation 1
Metrological Traceability and Accuracy in Microfluidic Devices: The Role of the MFMET II Project

Elsa Batista
Portuguese Institute for Quality
Head of volume and flow laboratory
Microfluidic devices are increasingly embedded in critical applications ranging from medical diagnostics and pharmaceutical development to environmental monitoring and advanced manufacturing. As these systems evolve toward ever smaller volumes and flow rates and critical measurements conditions, ensuring metrological traceability, measurement accuracy, and comparability of results has become a fundamental challenge. Despite significant technological progress, the lack of harmonized and standardized measurement procedures, and common calibration infrastructures continues to limit the reliability and interoperability of microfluidic technologies across laboratories and industrial sectors.
The EURAMET project MFMET II – Establishing Metrology Standards in Microfluidic Devices builds upon earlier foundational work to address these gaps by developing a coherent and internationally aligned metrological framework for microfluidics. The project focuses on establishing traceable methods for several metrological quantities such has flow resistivity, volume and flow velocity, designing and validating reference microfluidic devices, and improving uncertainty evaluation models appropriate for microscale fluid dynamics. Through collaborative research across European national metrology institutes, universities, and industry, MFMET II aims to generate robust calibration protocols, validated measurement techniques, and standardized procedures that directly support regulatory, research, and industrial needs.
A key objective of MFMET II is to bridge the current divide between conventional fluid metrology and microfluidic operation regimes by producing practical guidance and traceable calibration tools that can be readily adopted by manufacturers and end users. The project’s outcomes will enable consistent quality assurance, enhance reproducibility in research and industrial settings, and support emerging technologies that depend on precise fluid handling at the microscale. Ultimately, MFMET II contributes to shaping a sustainable, standardized metrology infrastructure that underpins innovation in microfluidics and strengthens European and global competitiveness in this rapidly expanding domain.
Brief Biography
Dr. Elsa Batista holds a PhD in Mechanical Engineering from Universidade Nova de Lisboa / Faculty of Sciences and Technology (FCT/UNL.
She has been responsible for the Volume and Flow Laboratory of the Portuguese Institute for Quality (IPQ) since 2002.
Dr. Batista serves as the convenor of the EURAMET Technical Sub Committee for Volume. She is also a board member of both the Microfluidics Association and the SPMET Association.
Since 2002, she has worked as a technical auditor and trainer in the fields of volume and flow. She is an active member of several international standardization committees, including ISO/TC 48, ISO/TC 28, ISO/TC 276, and ISO/TC 84, and currently chairs ISO/TC 48/SC 8.
In 2023, she received the CEN–CENELEC Standards + Innovation Award in the Individual Researcher category.
She is presently the coordinator of the EURAMET international project “MFMET – Establishing Metrology Standards in Microfluidic Devices II.”
Session I
Presentation 2
The Front-Track Method: A Metrological Standard to Measure Nanoflows

Kevin Romieu
CETIAT
Metrologist for the liquid flow laboratory
The accurate measurement of extremely low flow rates is essential for the development, calibration, and validation of microfluidic devices. While the gravimetric weighing method is a robust technique over a large range of flowrates, its use is increasingly challenging when trying to calibrate flow devices below 1 g/h.
To this end, optical methods are a helpful and efficient alternative to push accurate measurements down to a few nanoliters per minute. In particular, this talk focuses on the implementation of the front-track method as a primary standard for calibration. Its principle is to track the displacement of a meniscus—typically a water-air interface—inside a glass capillary of known diameter with a camera. Contrary to the weighing method, the reference quantity is a volume flow rate, instead of a mass flow. The talk presents how the front-tracking method is metrologically traced back to the International System of Units (SI) through length and time calibration of the reference equipment. It also gives a few good practices on the use of this method with examples from the MFMET project.
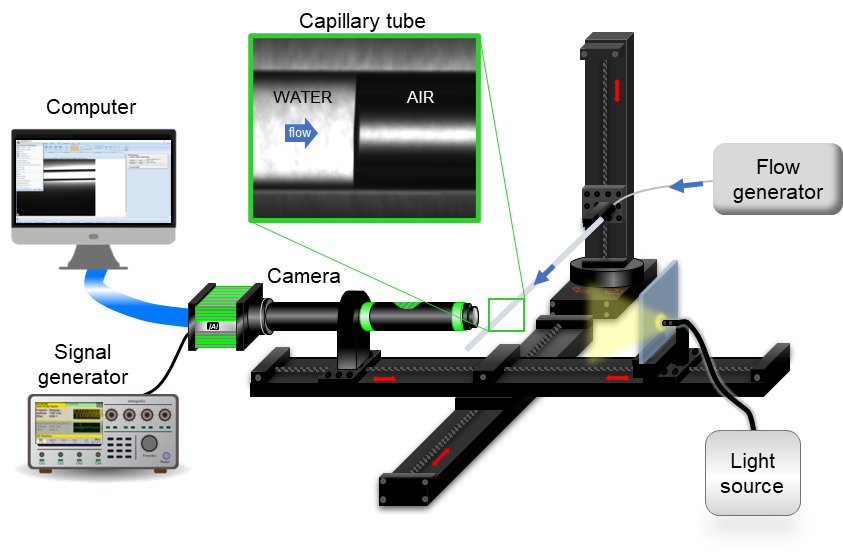
Brief Biography
Kevin Romieu holds a degree in Mechanical Engineering from the French “Grande école” Mines Saint-Étienne.
He has 10 years of experience as an R&D engineer for energy processes in various companies. In 2022, he joined CETIAT, a technical centre for the HVAC industry. It is also a designated institute by LNE, the French National Metrology Institute, for maintaining the primary standards for 3 quantities. Among these, Kevin is the metrologist in charge of the liquid flow laboratory.
Kevin is currently the convenor of the EURAMET technical subcommittee for Liquid Flow and a member of several standardization committees. He is also leading the work package 4 of the European project MFMET II “Establishing Metrology Standards in Microfluidic Devices”.
Session I
Presentation 3
Development of the calibration equipment for micro flowrate at National Metrology Institute of Japan

Shunsuke Kagami
National Metrology Institute of Japan (NMIJ)
Researcher
Microfluidic devices are widely utilized in various applications, including semiconductor manufacturing, chemistry, biology, and medicine. As these applications continue to expand, the demand for accurate calibration of micro flowrates is steadily increasing. In particular, there is a growing need for reliable calibration methods applicable to a wide range of liquids with different physical properties. This presentation focuses on the research activities of NMIJ, particularly the development of a calibration system that can serve as a primary standard and a microflow sensor that can be used as a transfer standard.
Brief Biography
Shunsuke Kagami received his master’s degree in engineering from the University of Tsukuba in 2023. He is currently working at National Metrology Institute of Japan (NMIJ). He is engaged in the calibration and development of measurement devices.
Session I
Presentation 4
Gravimetric Weighing: A Metrological Standard Method for Microfluidic Flow Measurement

Oliver Büker
RISE (Research Institutes of Sweden)
Senior researcher
The accurate measurement of extremely low flow rates is essential for the development, calibration, and validation of microfluidic devices. The gravimetric weighing method has become the primary metrological technique for microfluidic flow measurement, as it offers high accuracy, SI traceability, and minimal dependence on fluid properties. In microflow, this approach is based on measuring the increase in mass over time with a high-resolution weighing scale in the microgram range and subsequently calculating the flow rate. This calculation considers various physical corrections, such as density, temperature, pressure fluctuations, buoyancy effects, and evaporation rates. Consequently, the gravimetric method facilitates the reliable quantification of flow rates down to the nanoliter per minute range. This method is primarily used for the calibration and characterization of flow meters and infusion systems in metrology laboratories. It is considered a highly accurate reference standard and fundamental measurement method. Given that mass is an SI base quantity, gravimetric measurements provide a robust foundation for traceability in pharmaceutical dosing, biochemical research, and micro-reactor processes. As the method is independent of viscosity and optical properties, it is particularly advantageous in complex or heterogeneous media. Gravimetric weighing constitutes a fundamental and widely applicable approach for the validation of microflows and can also be used for the characterization of microfluidic chips. This technique offers a metrologically sound and experimentally flexible method for ensuring accuracy and reproducibility in microfluidic research and applications. The methodological framework for gravimetric weighing is formally established in ISO 4185:1980 and supplemented by current guidelines, including EURAMET guide CG-27 for the calibration of drug delivery devices and AAMI TIR101:2021 for the performance validation of infusion pumps. The adoption of a standardized approach facilitates the establishment of reliable metrological traceability and the quantification of uncertainty for the characterization of applications involving flow rates within the microfluidic range.
Brief Biography
Oliver Buker is a senior researcher at RISE Research Institutes of Sweden. He received his Ph.D. in Chemical and Process Engineering from the Technical University of Berlin in 2010 while employed as a researcher at PTB, the National Metrology Institute (NMI) of the Federal Republic of Germany. He then worked as a postdoctoral researcher at PTB for two more years, mainly focusing on flow measurements and laser optical measurement techniques. In 2012, he joined RISE, where he works in the Department of Measurement Science and Technology, which includes the NMI of Sweden for the physical quantities volume, flow, and density. Oliver has two decades of practical and theoretical experience from national, European, and international perspectives. He is a Swedish representative of the EURAMET Technical Committee for Flow (TC-Flow), BIPM CCM Working Group on Fluid Flow (WGFF), BIPM CCM Working Group on Density and Viscosity (WGDV), and IMEKO TC9 (Flow Measurement).
Session II
Presentation 1
Plastic-based Microfluidics and Nanofluidics: From Prototyping to Production

Steven A. Soper
The University of Kansas
Foundation Distinguished Professor
Delivery of microfluidic devices, especially those associated with clinical measurements, must go through several steps prior to translating into the market. These include prototyping through an iterative process to home in on a device concept optimal for the intended application, medium scale production for testing to secure the necessary analytical and clinical figures-of-merit for in vitro diagnostic applications, and finally large-scale production for commercial dissemination. In this presentation, I will present the fabrication techniques used to move through the production pipeline thermoplastic devices as the substrate of choice for generating microfluidic devices for in vitro diagnostics. Fabrication techniques that will be discussed for prototyping include high precision micromilling, which has a device turnaround time < 1 day and is a maskless process, and excimer laser machining. In terms of medium scale production, micromilling of embossing tools and the use of hot-embossing will be discussed to produce plastic devices for securing the figures-of-merit. Finally, the use of photolithography and Ni electroplating to form Ni mold inserts for high-scale production of devices for commercial applications using injection molding will be discussed. I will also talk about assembly processes that can be implemented to generate finished devices containing cover plates with high process yield rates. Following device assembly, I will discuss plastic surface modification strategies that can be implemented to allow for increasing surface wettability and biocompatibility as well as creating functional surfaces for biomolecule attachment. I will also share with participants how production lines can be employed to deliver nanofluidic devices, which possess fluidic vias <100 nm in dimensions and made from plastics. Finally, I will show some examples of plastic devices that went through the aforementioned production pipeline for clinical measurements, such as processing liquid biopsy markers for extracellular vesicles (see Figure 1). As an example of a nanofluidic device, a sensor consisting of 100 nm pores will be discussed to evaluate the full/empty status of adeno-associated viruses that are used for drug delivery (Figure 2).

Figure 1. Microfluidic device for the affinity selection of extracellular vesicles from plasma samples. The device is made from a plastic via injection molding and contains 1.5M micropillars.

Figure 2. Nanofluidic device for determining the full/empty status of adeno-associated viruses for drug delivery using single-particle resistive pulse sensing.
Brief Biography
Prof. Soper is a Foundation Distinguished Professor in Chemistry and Mechanical Engineering at the University of Kansas. He also holds an appointment at Ulsan National Institute of Science and Technology in Ulsan, South Korea, where he is a World Class University Professor. Prof. Soper is currently on the Editorial Board for Scientific Reports. As a result of his efforts, Prof. Soper has secured extramural funding totaling >$123M and has published over 265 peer-reviewed manuscripts (h index = 76; 19,985 citations) and is the author of 20 patents. He is also the founder of BioFluidica Inc., which is marketing microfluidic devices for the analysis of liquid biopsy markers. Prof. Soper has granted 68 PhDs and 7 MS degrees to students under his mentorship and currently heads a group of 20 researchers. He was recently inducted into the National Academy of Inventors.
Session II
Presentation 2
Material Design Criteria for Scalable Organ-on-Chip Platforms

Daniel Nawrocki
Kayaku Advanced Materials
CGTO
Organ-on-chip devices are commonly constructed from materials that are available rather than materials deliberately designed to meet the functional requirements of the biological system. This reflects the historical development of many platforms within microfluidic prototyping environments, where fabrication convenience and material accessibility often precede systematic evaluation of biological performance. As organ-on-chip technologies are increasingly positioned as enabling platforms for New Approach Methodologies (NAMs), the relationship between material choice and biological accuracy becomes increasingly important.
Organ-on-chip platforms are not single-material constructs but stacked-layer devices composed of structural substrates, membranes, surface coatings, and extracellular matrix interfaces. Each layer performs a distinct role within the system and therefore imposes different and often competing material requirements. Mechanical compliance, gas permeability, small-molecule interaction, optical performance, surface chemistry, bonding compatibility, dimensional stability, and manufacturability must therefore be evaluated as interdependent attributes across the architecture of the device rather than as isolated properties of a single material.
This work presents a framework for modeling the dependencies between these material attributes and for evaluating candidate materials using structured figures of merit associated with specific device roles within the layered architecture. The framework translates biological and engineering requirements into measurable material criteria and allows attributes to be weighted according to device architecture, assay objectives, and integration constraints. Materials are therefore compared not only individually but also in terms of how they interact within a layered device system.
Next-generation organ-on-chip platforms are therefore unlikely to converge on a universal construction material. They will require coordinated suites of materials optimized for different functional layers within the device. Systematic methods for comparing, qualifying, and integrating these materials provide a pathway toward more reproducible, scalable, and biologically credible organ-on-chip systems capable of supporting the performance expectations of NAM-based models.
Brief Biography
Dan Nawrocki is Chief Growth and Technology Officer at Kayaku Advanced Materials, where he leads initiatives in advanced polymer materials for semiconductor microfabrication, microdevices, and emerging organ-on-chip platforms. His work focuses on translating polymer chemistry and materials engineering into manufacturable technologies used in lithography, MEMS fabrication, and next-generation biomedical systems.
Over the course of his career, Nawrocki has worked extensively in specialty materials development, process scale-up, and microfabrication technologies. Prior to joining Kayaku Advanced Materials, he held technical and leadership roles at MicroChem Corporation and earlier worked in coating science and process development at Rohm and Haas and InteliCoat Technologies.
He is an inventor on multiple patents related to photoimageable polymers, dielectric materials, and advanced microfabrication processes. His recent work focuses on engineered polymer systems that enable scalable organ-on-chip devices and strengthen the material foundations of next-generation in vitro testing platforms.
Session II
Presentation 3
Standardization of Microfluidic Devices
– Indicators for plastic material selection –

Keisuke Shiota
Zeon corporation, Research & Development Center, Specialty Plastics Laboratory
Researcher
Microphysiological Systems(MPS) not only enables highly accurate assessment of drug toxicity and safety, but is also attracting increasing attention as an alternative to animal testing; it is anticipated that a variety of products will continue to be developed to facilitate diverse forms of evaluation. Whilst a wide range of materials are currently used in MPS devices, it is desirable that the appropriate material be selected according to the intended application. To determine which materials are suitable for a device, it is necessary to verify whether the material’s properties align with the intended application. However, there is currently no internationally agreed-upon document defining what physical properties are suitable for MPS devices or how these properties should be evaluated, and there are no guidelines whatsoever on how to select materials.
We are therefore working to develop an international standard to serve as a guideline for material selection, so that developers and users of MPS devices can carry out the required evaluations using appropriate materials. In this presentation, we will focus on the physical properties required of materials for MPS devices and the methods for evaluating them, whilst providing an overview of the standard currently under development.
Brief Biography
Keisuke Shiota has been working at Zeon corporation since 2022, where he is engaged in research into the adsorption of biomaterials onto resin materials and the development of resins for bio-devices. He also serves as project leader for the proposal currently under consideration in TC 48/WG 3, entitled ‘Microfluidic devices — Indicators for material selection used for microfluidic devices’.
Session III
Presentation 1
Integrated sensors and actuators for microfluidic handling systems

Joost Lötters
1) Bronkhorst High-Tech B.V., The Netherlands
Science Officer
2) University of Twente, The Netherlands
Professor of Microfluidic Handling Systems
The growing complexity of modern microfluidic applications requires a fundamental shift from passive fluid channels to active, intelligent systems. For processes such as precisely administering medication through multi-infusion, real-time chemical synthesis in micro reaction systems and cell culture growth and monitoring in organ-on-a-chip systems, there is a need for integrated sensors and actuators that can measure and adjust critical parameters such as flow rate, pressure, viscosity and relative permittivity with high accuracy.
The integration of sensors directly into the microchannels is essential for their performance. Using surface channel technology, many different microfluidic transducers can be integrated on the same chip. Examples of fabricated devices are a piezo-electrically actuated proportional control valve, thermal and Coriolis flow sensors, pressure sensors, thermal conductivity sensors, and relative permittivity sensors.
Additional parameters can be determined by finding correlations between sensor signals. For instance, viscosity can be derived from the relationship between pressure drop and flow rate through the Hagen-Poiseuille equation. Furthermore, machine learning techniques can be explored to find “hidden” correlations between physical properties—such as the link between glucose concentration and viscosity, or pH and conductivity.
To translate these measured values into action, integrated actuators are indispensable. Mechanical components such as micropumps and microvalves facilitate the development of closed-loop feedback systems.
Ultimately, the integration of sensors and actuators may lead to compact, autonomous lab-on-a-chip type systems that are capable of delivering laboratory-quality results in a miniaturized format. This technological convergence is driving the next generation of microfluidic instrumentation for life sciences and industrial process control.
In the lecture, examples of integrated microfluidic sensors and actuators will be presented, as well as their performance in several different applications, including organ-on-a-chip, multi-infusion systems, and flow chemistry. Some commercially available products resulting from the research will be shown, and an outlook on future research will be given.
Brief Biography
Prof. dr. ir. Joost C. Lötters received the M.Sc. in 1993 and Ph.D. degree in 1997 in electrical engineering from the University of Twente, The Netherlands. In 1997, he joined Bronkhorst High-Tech BV, The Netherlands, where his research focused on thermal, Coriolis and ultrasonic flow sensing. In 2015, he was appointed part-time professor microfluidic handling systems at the University of Twente. Since then, his research has focused on integrated systems for the accurate measurement, control, dosage and analysis of (micro) flows. Joost Lötters is (co-)inventor of more than 30 patents and (co-)author of more than 120 journal and conference papers.
Session III
Presentation 2
Next-Generation Organ-on-a-Chip Platforms with Integrated Sensors for Dynamic Cell Measurements

Darwin R. Reyes
National Institute of Standards and Technology (NIST)
Project Leader
Microphysiological Systems offer features such as well-controlled cell microenvironments, allowing the introduction of chemical, biological, and physical stimuli. However, with a few exceptions, these platforms use endpoint readouts, where results are obtained at the end of the experiment. At this point, cell responses are analyzed at specific time points. Endpoints require a higher throughput when compared to an approach that can continuously measure cell responses in real-time. Furthermore, important information regarding the effects of drugs and toxicants is lost when endpoint measurements are used. Thus, the integration of sensors within these platforms enables continuous monitoring of cell responses to stimuli, which in turn provides a complete dynamic picture of cell behavior, instead of a snapshot in time. This work presents the development of a platform, which integrates electrodes on both sides of porous polyester membranes to measure electronically and optically cell behavior, such as migration and cardiomyocytes (heart cells) beating. This platform is currently used for Heart-on-Chip research as well as for quantitative cancer cell migration assays. Multiparametric measurements are also performed with these platforms for different functionalities such as barrier integrity and changes in the frequency and amplitude of heart cells beating.
Brief Biography
Dr. Darwin R. Reyes is a project leader in the Microsystems and Nanotechnology Division of the Physical Measurement Laboratory (PML) at the National Institute of Standards and Technology (NIST), and a Lecturer at the Division of Biotechnology at Johns Hopkins University. His work at NIST has focused on the development of electronic methods for manipulating and measuring cells in cell-based bioassays. More recently, he has been developing tissue/organ-on-a-chip devices with integrated electronic and optical measurement tools to enable real-time Heart-on-a-Chip and cancer cell-based testing for drug development and toxicity assays.
Dr. Reyes is a Co-founder and Chair of the Microfluidics Association (MFA), an international standards development organization that fosters the development of ISO standards and guidelines for the microfluidics community. He is also the Chair of the International Micro Physiological Systems (IMPS) Society’s Standardization Interest Group. Dr. Reyes was recently bestowed with the American Institute for Medical and Biological Engineering’s (AIMBE) Emerging Leader Award and the PML/NIST Outstanding Achievement in Measurement Services and Standards Award for his contributions to the development of ISO standards through his work with the MFA and other stakeholders.
Session III
Presentation 3
Development and Optimization of a Stirrer-Based On-Chip Micropump for Microfluidic devices

Hiroshi Kimura
Tokai University
Professor
Microphysiological systems (MPS) have attracted considerable attention as alternatives to animal testing in drug development by recapitulating organ functions and organ–organ interactions using microfluidic device technologies. Conventionally, perfusion control in microfluidic devices relies on external pumping systems such as peristaltic pumps, syringe pumps, and pneumatic pumps. However, these systems require external power sources, complex tubing networks, and bulky control units, which hinder system miniaturization and integration.
In this study, we propose an on-chip micropump (stirrer-based micropump) that eliminates the need for external tubing, thereby fundamentally addressing these limitations. This approach utilizes the rotating magnetic field of a magnetic stirrer to non-contactly drive a stir bar integrated within a microchannel pump chamber, thereby enabling fluid perfusion. This design allows complete integration of the pump and microfluidic device, significantly reducing design constraints associated with flow control in conventional microfluidic platforms.
To overcome the previously reported issue of flow instability, we systematically optimized both the pump structure and operational parameters. As a result, we demonstrate that stable and reproducible fluid transport can be achieved. In this presentation, we report the design principles and optimization strategies of the proposed micropump and discuss its potential as a novel flow control platform for highly integrated and simplified microfluidic devices.
Brief Biography
Dr. Hiroshi Kimura is a Professor at the Micro/Nano Technology Center, Tokai University, where he has been since 2012. He received his Ph.D. in Bioengineering from the University of Tokyo in 2007. From 2007 to 2012, he worked at the Institute of Industrial Science (IIS), University of Tokyo, where he served as a Project Assistant Professor. During 2017–2018, he was a Visiting Research Fellow at the University of California, Los Angeles (UCLA). His research focuses on microfluidic device and system technologies and their applications in biological sciences, particularly microphysiological systems.
Session IV
Presentation 1
Open Discussion: “Who is leading the MPS project with AI?”


Moderators: Darwin R. Reyes and Hiroki Nakae
This session aims to highlight the “knowledge gap” existing between wet-lab researchers and AI experts, exploring paths to bridge this divide through a floor-wide discussion. Centered on the pivotal question, “Who is leading the MPS project with AI?”, we will engage in a 30-minute interactive dialogue with all participants.
Session IV
Presentation 2
Data Integration and Curation for AI-Driven Drug Discovery

Kenji Mizuguchi
Institute for Protein Research, The University of Osaka
Professor of Computational Biology
Advances in artificial intelligence (AI) critically depend on the availability of a large amount of high-quality public data in a computer-friendly format. In many biological domains, however, including disease biology and drug discovery research, the limited availability of data and insufficient integration and curation remain major barriers to effective AI utilization.
To address this issue, we have developed DruMAP (Drug Metabolism and pharmacokinetics Analysis Platform), an online system consisting of a curated database of drug metabolism and pharmacokinetics (DMPK) parameters for more than two million chemical compounds and a web interface to a range of predictive models, built based on those data (Kawashima et al, J. Med. Chem. 2023).
By capitalizing on this and other related achievements, we argue, in this lecture, that systematic data organization is a critical foundation for the next generation of AI-driven science. Using pharmacokinetic (PK) modeling as a central example, we explore strategies to overcome data limitations and enhance model development. Specifically, we discuss: (1) the potential to generate new, task-relevant data from related datasets even when direct measurements are unavailable; (2) the use of generative AI and AI agents to extract, curate, and integrate information from heterogeneous and fragmented sources; and (3) the application of transfer learning, multitask learning, and hybrid modeling to compensate for sparse data and improve predictive performance.
Through these perspectives, we highlight how advances in data integration and curation can enable more robust and scalable AI-driven PK modeling, ultimately accelerating drug discovery and development.
Brief Biography
Dr. Kenji Mizuguchi is Professor of Computational Biology at Institute for Protein Research, The University of Osaka. His research focuses on the development of computational methods for predicting protein structure, function and interaction, as well as data integration and database development, all aiming to establish systems approaches to drug discovery. He was previously Director of Artificial Intelligence Center for Health and Medical Research (ArCHER), at National Institutes of Biomedical Innovation, Health and Nutrition, Japan, a University Lecturer at the Department of Applied Mathematics and Theoretical Physics, and a Group Leader at the Department of Biochemistry, University of Cambridge. He received a BA degree in Physics in 1990 and a PhD degree in Chemistry in 1995, both from Kyoto University.
Session IV
Presentation 3
Integrated Knowledge Graph and Domain-Specific Language toward Compound Toxicity Assessment in Microphysiological Systems

Hitoshi Fujimiya
Dynacom Co., Ltd.
Chairperson
This presentation describes the development of a computational infrastructure for compound toxicity prediction in Microphysiological System (MPS) research. Effective utilization of experimental data from Organ-on-a-Chip platforms requires systematic integration of accumulated large-scale omics information into a unified framework for toxicity prediction and drug efficacy evaluation.
We constructed an integrated knowledge graph using Neo4j by collecting and linking 12 major biomedical data sources: ChEMBL (2.9M compounds), LINCS L1000 (473K gene expression signatures), STRING (protein-protein interactions), CoxpresDB (3.1M co-expression pairs), Reactome (2,786 pathways), PharmGKB, AOPWiki (391 adverse outcome pathways), ToxCast (9,400+ chemicals), ChEBI, RefMet, and chemical toxicity databases extracted using large language models. The resulting knowledge graph comprises approximately 4.4 million nodes and 455 million relationships.
To enable stable and reproducible querying of this deeply hierarchical database, we developed Morpheus DSL, a domain-specific language grounded in category theory. Node types (compounds, proteins, genes, pathways) are defined as categorical objects and their relationships as morphisms, ensuring type safety and composability. The system operates through a multi-stage pipeline from natural language input to automated report generation. In practice, AI generates Morpheus syntax, so researchers do not need to memorize the language — the example on the slide illustrates the structure.
Morpheus DSL enables multi-layered compound searches across structural similarity, protein targets, biological pathways, gene co-expression networks, and adverse outcome pathway analysis. We demonstrated its utility through drug repositioning analyses. For toxicity prediction, we have been integrating established published methods — including ADMET-AI, physicochemical descriptors, and bioactivity features — into a Drug-Induced Liver Injury (DILI) prediction pipeline as an initial step. Evaluation using publicly available datasets is ongoing. Looking ahead, we are developing a two-stage prediction architecture combining Graph Neural Networks and Bayesian network-based reasoning along AOP causal chains, with the goal of organically connecting this framework with MPS Organ-on-a-Chip data toward organ-specific toxicity risk estimation.
Brief Biography
Hitoshi Fujimiya began his career within the Hitachi Group, where he spearheaded the development of pioneering hardware, including fluorescent bio-image analyzers for DNA sequence analysis and high-speed, high-precision homology search LSIs. In 1995, he founded Dynacom Co., Ltd. to bridge the gap between advanced engineering and the life sciences. Since then, he has led the development of sophisticated software solutions for DNA sequence clustering and disease-related SNP analysis.
In recent years, he has focused on integrating clinical data with high-performance computing, successfully constructing predictive models for COVID-19 severity based on blood cytokine dynamics. Currently, he is directing research and business initiatives centered on genomics and multi-omics data analysis, integrating AI to advance life science applications. With a foundation in world-class industrial R&D and a decades-long track record in bioinformatics, Hitoshi Fujimiya continues to drive innovation at the intersection of AI, genomics, and personalized medicine.
Session V
Presentation 1
How standardization is addressed by companies

Carlo Alberto Paggi
Chiron
Managing Director
Organ-on-chip (OoC) technologies are increasingly recognized as powerful tools for generating human-relevant data in preclinical research, yet their broader adoption depends on how effectively companies address the question of standardization. In this lecture, Carlo Alberto Paggi will discuss how organ-on-chip developers approach standardization at multiple levels, from the hardware itself to the intended biological application. Particular attention will be given to the distinction between technical standardization—such as geometry, sterility, manufacturability, and operational consistency—and application-oriented standardization, which concerns robustness, reproducibility, and fitness for a specific research or industrial question.
The lecture will also examine why standardization should not be conflated with qualification and validation in a regulatory context. While industrial and ISO-oriented standardization can support comparability and confidence across platforms, regulatory acceptance for drug development requires a different lens. In that setting, qualification and validation depend on context of use, evidentiary strength, and the expectations of regulatory bodies regarding safety, toxicology, pharmacokinetics, efficacy, and mechanism-of-action studies. The presentation will therefore outline distinct routes by which OoC technologies may enter the regulatory process: either as supporting data within an existing dossier or through the validation of a platform for a defined application.
Drawing on company experience with advanced in vitro systems such as mechanically active tissue platforms and microplate-based organoid systems, the lecture will highlight how developers are translating innovation into credible and actionable data packages for industry. Using examples from disease modelling and preclinical development, it will argue that the future of OoC adoption lies not only in technological sophistication, but in building standardized, qualified, and application-relevant systems that can meet both industrial needs and regulatory expectations. This keynote is positioned within the symposium’s session on “Standardization and regulation,” underscoring its relevance to the broader microfluidics community.
Brief Biography
Carlo Alberto Paggi, PhD, is a scientist and entrepreneur working at the intersection of biomedical engineering, organ-on-chip technologies, and animal-free innovation. With an academic background that includes studies at the University of Twente and research focused on tissue engineering, cartilage biology, and mechanobiology, he has contributed to the development of advanced in vitro models for joint disease research. He is also publicly associated with chiron as a co-founder and leadership team member, where his work centers on translating cutting-edge organ-on-chip science into practical applications for arthritis, cancer research and drug development
Session V
Presentation 2
Characterization and Standardization of Organ Cells for MPS – Prospects for the Development of ISO Standards

Yuzuru Ito
Institute of Life and Environmental Sciences, University of Tsukuba
Professor
Microphysiological systems (MPS), a kind of NAMs, are complex in vitro models that integrate cells and devices, and are expected to be utilized in various phases of the drug discovery process. In recent years, there has been a growing demand for robust MPS operations, which requires cooperative optimization of organoids and devices. This presentation will focus on the characterization of organ cells and the standardization of evaluation method development processes using ISO documents, and will discuss approaches to achieve the social implementation of MPS.
Brief Biography
Prof. Yuzuru Ito is a specialist in stem cell biology and biotechnology, with a particular emphasis on developing advanced drug discovery support platforms, including microphysiological systems (MPS). He received his B.Sc. in Biology from the University of Tokyo in 1994 and completed his Ph.D. in Science at the same institution in 2001. Following research appointments at Kyushu University and the University of Tokyo, he served as Group Leader in the JST ICORP Organ Regeneration Project, contributing to foundational advances in organogenesis and regenerative biology. He subsequently led research in organ development and stem cell engineering at the National Institute of Advanced Industrial Science and Technology (AIST) until his departure in 2020. Since 2020, Prof. Ito has held a professorship in the Life and Environmental Sciences program at the University of Tsukuba. He also contributes to life science innovation at Chiyoda Corporation, promoting translational research and industry–academia collaboration.
Session V
Presentation 3
Standardization of MPS in ISO

Yutaka Yanagita
Forum for Innovative Regenerative Medicine
Secretariat
In this lecture, I will report on the history and status of MPS standardization within ISO, focusing on the establishment of the collaborative framework between ISO/TC 48 and ISO/TC 276, and discuss future developments.
Brief Biography
1984Ph.D. in biochemistry, University of Tokyo
1984-1986Postdoctoral Fellow, Cornell University
1986-1987Research Assistant, Nagasaki University
1987-2022Astellas Pharma
2013-Forum for Innovative Regenerative Medicine
Expert, ISO/TC 276/WG 4 and other WGs
2024-Expert, ISO/TC 48/WG 3








